Distillation Tutorial II: McCabe-Thiele Method of Distillation Design (Main Page)
The McCabe-Thiele method of distillation design is a graphical method of design that dates back to the early 20th century. It is used as an introductory method to teach the design of distillation and is still in use today by engineers who design distillation columns. The McCabe-Thiele method considers binary distillations but can also be used for what are called pseudo-binary mixtures (i.e. mixtures that are treated as binary by identifying light and heavy key components).
1. Introductory Concepts. The main features of the McCabe-Thiele method are that it:
- Combines equilibrium with mass balance concepts.
- Is a graphical approach.
- Applies to (pseudo-) binary mixtures.
- Uses constant molar overflow (CMO) assumptions.
- Approximates energy balance effects and energy demands.
Binary Distillation. The easiest way to understand the McCabe-Thiele method is graphically. To do this, we begin with a slightly more detailed schematic of a distillation column than shown in Fig. 1 of Tutorial I.
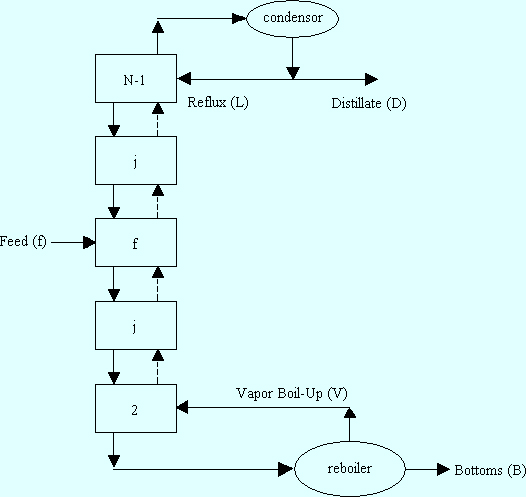 |
2. The McCabe-Thiele Method. One of the key features of the McCabe-Thiele method (as well as other distillation design methods) is that it is assumed that vapor entering from the stage below and liquid entering from the tray above come in contact with each other and leave any given stage in equilibrium. Before presenting the equations of the McCabe-Thiele method we illustrate it graphically for a 50-50 % mixture of saturated liquid ethylene and ethane at 100 psia. We will call this column a C2 splitter. Finally, we specify the reflux ratio to be r = 3.9. We suggest that you follow each step by referring to Fig. 2 below.
The Graphical Steps of the McCabe-Thiele Method
- Draw the yx diagram for the binary mixture of interest at the operating pressure of the distillation column. Typically y and x refer to the mole fraction of the light component.
- Draw the 45-degree line.
- Draw the feed line based on the quality (or energy content of the feed). For saturated liquid, this is a vertical line running from the feed composition through the equilibrium curve. For saturated vapor, this line is a vertical line from the feed composition to the 45-degree line, then a horizontal line from the 45-degree line through the equilibrium curve. Other feed conditions can also be drawn with the aid of a simple equation and will be discussed later in this tutorial in the section on feed quality.
- Place the desired bottoms composition, xB, and the desired distillate composition, xD, on the x-axis.
- For a specified reflux ratio, r, draw the 'rectifying'
line. This rectifying line begins at the point xD on the
45-degree line and has a slope of
r / (r+1). - Draw the 'stripping' line by connecting the intersection of the feed line and the rectifying line and the point xB on the 45-degree line. The slope of the stripping line is related to r because r fixes the point of intersection of the rectifying line and the feed line. This intersection and xB uniquely fix the stripping line and thus its slope. We usually denote the slope of this line by (s+1) / s where s is the vapor boil-up ratio. In this example, s works out to be 4.5.
- Beginning at the point xD on the rectifying line, draw a horizontal line to the equilibrium curve and then a vertical line to the operating (rectifying or stripping) line.
- Repeat step 7 forming a staircase until you reach or pass the point xB on the 45-degree line.
- Each point where the staircase intersects the equilibrium curve denotes one stage in the column.
We can also begin stepping off stages in any distillation column by starting at the point xB on the 45-degree line and moving upward. It is also possible to specify a desired boil-up ratio, s, instead of a desired reflux ratio, r.
Practice Exercises. Understanding the graphical nature of the McCabe-Thiele method is an important first step in understanding distillation design. Consider a 30-70 % mixture of ethane and propane at 150 psia where the feed is saturated liquid at its bubble point. Suppose the desired bottoms composition is xB = 0.05 and the desired distillate composition is xD = 0.99. Let the boil-up ratio be specified at s = 2. Answer the following:
- Use the McCabe-Thiele graphical procedure and determine the reflux ratio r.
- Determine the number of equilibrium stages that are required for this distillation.
- Compute the temperatures of the feed, bottoms, and distillate by determining their respective bubble points.
Just press play.
3. Equations of the McCabe-Thiele Method. There are a number of important equations that come from the McCabe-Thiele method that are related to mass and energy balancing. These equations include the operating lines (i.e. rectifying and stripping lines), the feed line, the relationship between reflux ratio and boil-up ratio, and the determination of heating and cooling requirements.
Mass Balance Considerations. Mass balancing is an important part of distillation column design, analysis, and simulation. Suppose we return to our C2 splitter illustration, which is fed with 100 lbmol/h of a 50-50 % mixture of ethane and ethylene, has a feed that is saturated liquid at its bubble point, and operates at 100 psia. The desired composition of the bottoms product is xB = 0.05 and the composition of the distillate is xD = 0.98. We now illustrate how mass balancing determines the rectifying and stripping lines in the McCabe-Thiele method.
Operating Lines. Go back to Fig. 1 and draw an imaginary (mass balance) box that encloses the reboiler and all stages up to and including stage j in the bottom section of the column so that only the bottoms product and the streams entering or leaving stage j cross this imaginary box. This imaginary box is often called a mass balance envelope. If we now write a total mass balance for this imaginary box we get
| Lj+1 = Vj + B | (2.1) |
If we also write a component mass balance for ethylene, which we will call component 1, we get
| Lj+1xj+1 = Vjyj + BxB | (2.2) |
Let's solve Eq. 2.2 for yj. This gives
| yj = [Lj+1 / Vj] xj+1 -½ [B / Vj] xB | (2.3) |
Now let's use Eq. 2.1 to eliminate Lj+1 from Eq. 2.3. To do this simply substitute Vj + B for Lj+1 in Eq. 2.3. This gives
| yj = [(Vj + B) / Vj] xj+1 -½ [B / Vj] xB | (2.4) |
If we define s, the stripping or boil-up ratio, to be s = V/B, then Eq. 2.4 can be re-written as
| yj = [1 + B / Vj] xj+1 -½ [B / Vj] xB = [1 + 1 / s] xj+1 -½ [1 / s] xB | (2.5) |
Finally, we can substitute s/s for 1 in the first term on the far right of Eq. 2.5 and get
| yj = [(s + 1) / s] xj+1 -½ [1 / s] xB | (2.6) |
which is called the stripping line. Now it is important for you to see that the slope of this line is (s+1)/s and that it intersects the 45-degree line at the point xB. First, we consider Eq. 2.6 to define yj as a function of xj+1. Thus Eq. 2.6 can be written in the familiar form y = mx + b. If you then compare Eq. 2.6 to y = mx + b, the slope is (s+1)/s. Understanding why Eq. 2.6 intersects the 45-degree line at xB is a little harder. Here you must realize that intersecting the 45-degree line at xB means yj = xj+1 = xB. So if we set yj = xB in Eq. 2.6 and do some simple algebra we should get that xj+1 = xB.
Let's do that.
| xB = [(s + 1) / s] xj+1 -½ [1 / s] xB | (2.7) |
| [1 + 1/s] xB = [(s + 1) / s] xj+1 | (2.8) |
However, from Eq. 2.5 we know that the last term in Eq. 2.8 can also be written as [1 + 1/s]xj+1. Therefore, Eq. 2.8 can be written as
| [1 + 1/s] xB = [1 + 1/s] xj+1 | (2.9) |
By dividing both sides of Eq. 2.9 by [1 + 1/s], we see that xj+1 = xB.
Practice Exercises. To strengthen your mass balancing skills, answer the following.
- Mimic the analysis we have just done to show that the
equation for the rectifying line in the top section of the distillation
column is
yj = [r / (r+1)] xj+1 + [1 / (r+1)] xD, where r = L/D. - Show that this rectifying line intersects the 45-degree line at xD.
Energy Considerations in the McCabe-Thiele Method. Remember we said that distillation can use large amounts of energy. This energy is supplied in the form of heat to the reboiler and cooling to the condenser. Although it's not obvious, the McCabe-Thiele method does take into account energy considerations in an approximate way through the boil-up and reflux ratios s and r respectively. The definition of s is s = V/B where B is the bottoms product flow rate and V is the flow of vapor from the reboiler. Using a total mass balance around the reboiler (see Fig. 1) we see that the total amount of liquid material entering the reboiler is
| L = V + B | (2.10) |
Since liquid bottoms B and vapor V both leave the reboiler, V represents the amount of entering liquid L that is vaporized by adding heat. We can express this portion in terms of the boil-up ratio s as follows. Dividing both sides of Eq. 2.10 by V we get
| L/V = 1 + 1/s = (s + 1) / s | (2.11) |
Rearranging Eq. 2.11 we get
| V = L[s / (s+1)] | (2.12) |
which shows that V is just L multiplied by the fraction s/(s+1). Note that s/(s+1) must lie between 0 and 1. For s = 0, there is not vapor flow since Ls = 0 and this is consistent with the fact that s = V/B = 0 means V = 0. For s = 0.5, the amount of vapor is V = [0.5/1.5]L = L/3 . This says the vapor flow is one third that of the liquid flow to the reboiler when s = V/B = 0.5. Note that it also says (by mass balance) that B = 2V. Double checking these results with the overall mass balance for the reboiler shows that
| L = V + B = L/3 + 2V = L/3 + 2L/3 = L |
so everything is fine.
As s increases, the amount of vapor increases from a minimum of 0 when s = 0 to a maximum of L when s = infinity, s/(s+1) = 1, and B = 0.
Now that we have a way to calculate the vapor rate V, we can also compute the heat energy needed to generate that vapor. To calculate the heat required, we need to first determine the heat of vaporization of the bottoms product. To do this, we need the heats of vaporization of both components in our mixture and we must also use a very common engineering approximation of weight averaging. Suppose we denote the heat of vaporization of ethylene in our example by l1 and the heat of vaporization of ethane by l2. The bottoms product has a composition xB, where xB is the mole fraction of ethylene since it is the light component. Thus the mole fraction of ethane in the bottom product is (1 -½ xB). Calculating the heat of vaporization of the bottoms product by weight averaging means that we use the following simple formula:
| λB = xB λ1 + (1 -½ xB) λ2 | (2.13) |
where λB is used to denote the heat of vaporization of the bottoms product. The amount of heat required in the reboiler, which we denote by QR, can now be calculated using the equation
| QR = VλB | (2.14) |
We can also determine the amount of cooling required in the condenser in much the same way that we just determined the heating requirements for the reboiler. To do this we can simply use the weight averaging formula
| λD = xD λ1 + (1 -½ xD) λ2 | (2.15) |
where here λD is the heat of vaporization of the distillate product, xD is the composition of ethylene in the distillate product, and (1 -½ xD) is the composition of ethane in the distillate. The amount of cooling can be calculated using the formula
| QC = VλD | (2.16) |
where V is the amount of vapor entering the condenser or the vapor from stage N-1. Note that this assumes that all of the vapor stream entering the condenser is condensed and then split into liquid reflux and liquid distillate.
Practice Exercises.- Calculate the heat of vaporization of the bottoms and distillate products for the C2 splitter example using Eqs. 2.13 and 2.15.
- Determine the amount of heat required in the reboiler for a specified stripping ratio of s = 4.5.
- How much cooling is required in the condenser if r = 3.9?
Energy Balances and CMO Behavior. There is a subtle but important energy consideration that is associated with constant molar overflow.
Stages Other Than Feed Stages. For any distillation stage, say stage j, without an external feed stream, CMO implies that
| Lj+1 = Lj | (2.17) | |
| Vj-1 = Vj | (2.18) |
These equations result by drawing an imaginary energy balance envelope around any single stage in the rectifying or stripping sections of the distillation column -½ other than a feed stage -½ as shown in Fig. 3 in combination with some physical approximation.
| Lj+1 | Vj |
 |
| Lj | Vj-1 |
Equations 2.17 and 2.18 can be interpreted physically by saying that CMO means that the heats of vaporization of both components are equal. What this also means is that the amount of vapor condensed at stage j is exactly balanced by the amount of liquid boiled at stage j resulting in no net change in liquid or vapor entering and leaving stage j. Now this is not necessarily a good assumption and can cause students to question the reliability of the McCabe-Thiele method. However, in many cases the CMO assumption provides a reasonable approximation.
Quick Exercises. Determine how reasonable the CMO assumption is by answering the following:
- Calculate λB and λD for the C2 splitter illustration.
- What is the percentage difference in these values?
- Repeat questions 1 and 2 for acetone and water assuming the same product compositions as were used in the C2 splitter.
Feed Stages. The energy balance considerations around any stage that has an external feed must take into account the thermal quality (or energy content) of the feed stream. By this we mean that the energy content of the feed could be any of the following:
- Saturated liquid at its bubble point.
- Saturated vapor at its dew point.
- A mixture of vapor and liquid (see the Txy diagrams from Tutorial I).
- Sub-cooled liquid at a temperature below its bubble point temperature.
- Super-heated vapor at a temperature above its dew point temperature.
We use the symbol q to denote the feed quality, where q = LF/F and is the fraction of feed that is liquid. Thus immediately we see that for saturated liquid, q = 1. For saturated vapor, q = 0. For a mixture of liquid and vapor feed, 0 < q < 1. For sub-cooled liquid feed, q > 1 and for super-heated vapor feed, q < 0.
- Saturated Liquid Feed. For saturated liquid feed at
its bubble point temperature, approximate energy balance considerations
with the CMO assumption given the following equations.
Lj+1 + F = Lj (2.19)
Vj-1 = Vj (2.20) - Saturated Vapor Feed. For vapor feed at its dew
point we get a different set of equations resulting from energy balance
approximation. Here the equations are
Lj+1 = Lj (2.21)
Vj-1 = Vj + F (2.22) - A Mixture of Vapor and Liquid Feed. In this case, we
must either know the feed quality or determine it. Nonetheless, since 0
< q < 1, we get the following approximate energy balance results for a
feed that is a mixture of liquid and vapor.
Lj+1 = Lj + qF (2.23)
Vj-1 = Vj + (1 - q)F (2.24) - Sub-Cooled Liquid Feed. To determine the heat
effects of sub-cooled liquid, we must know how sub-cooled the liquid is;
therefore,
we must know its temperature. Once we know the feed temperature, we can
then compare it to the bubble and dew point temperatures of the feed to
calculate a value of q using the formula
q = 1 -½ [TF -½ Tbp] / [Tdp -½ Tbp] (2.25)
where TF is the temperature of the sub-cooled liquid feed. This equation is correct since for any sub-cooled liquid feed, TF < Tbp and therefore the second term in Eq. 2.25 is actually positive making q > 1. - Super-Heated Vapor Feed. Equation 2.15 can also be used for determining q for any super-heated vapor. Note in this case, TF > Tdp and therefore the second term in Eq. 2.25 is actually negative and less than -1 making q < 0.
Now it is important to understand that Eqs. 2.23, 2.24, and 2.25 actually apply to all five cases of feed quality. This is nice because then only one set of equations can be used to perform approximated energy balance calculations and their impact on the changes in liquid and vapor flow at any stage with an external feed.
Practice Exercises. In order to better understand Eqs. 2.23, 2.24, and 2.25, answer the following:
- For the C2 splitter example, determine the temperature of the saturated liquid feed. What do you get for a value of q using Eq. 2.25?
- Suppose this feed was sub-cooled by 1.5 K. What would be the value of q?
- Suppose the feed was saturated vapor. What would be the value of q?
- What is the thermal quality of a feed that is at a temperature halfway between its bubble point and dew point temperatures?
- For a vapor feed that is 20 K super-heated, what is the value of q?
q or the Slope of the Feed Line. To connect the previous discussions of approximate energy balance, thermal quality of the feed and the McCabe-Thiele Method, we need to show that Eq. 2.25 together with the definition of q = LF/F can be placed on a McCabe-Thiele plot and make sense. For this we need to understand that q is related to the slope of the feed line. This calculation of q is very convenient and allows us to draw the feed line properly from the intersection of a vertical line drawn from the feed composition and the 45-degree line.
Mathematically, the slope of the feed line is given by the simple equation
| q / [q -½ 1] | (2.26) |
If you think about this for a minute and go back to your calculations for q, you will see that
- For a saturated liquid feed, q = 1 and the slope of the feed line is infinity. The feed line is a vertical line.
- For saturated vapor feed, q = 0 and the slope of the feed line is also zero. The feed line is horizontal and goes toward the equilibrium line in this case.
- For a mixture of liquid and vapor, 0 < q < 1 and the slope of the feed line is negative. The feed line is a line between vertical and horizontal with a negative slope that goes toward the equilibrium line.
- For sub-cooled liquid, q > 1 and the slope of the feed line is positive but less than infinity.
- For super-heated vapor, q < 0 and the slope of the feed line is also positive.
Quick Exercise. For the results from the last set of practice exercises, draw the feed line for saturated liquid, saturated vapor, a mixture of liquid and vapor, sub-cooled liquid, and super-heated vapor feed.
Energy Balance Relationship Between Reflux Ratio and Boil-up Ratio. Using an energy balance around the entire distillation column, sometimes called an overall energy balance, it is possible to show that the reflux ratio r and the boil-up ratio s are related by the following equation
| s = (r + q)[xF -½ xD] / [xB -½ xF] + q -½ 1 | (2.27) |
Thus the amount of vapor generated in the reboiler is tied to the amount of vapor condensed in the condenser, which makes sense.
Summary Practice Exercises. Answer the following questions in order to practice what you have learned in this second tutorial by considering the separation of propylene and propane (a C3 splitter) at 200 psia. Let the feed be a 50-50 mol % mixture of saturated vapor and have a flow rate of 100 lbmol/h, xD = 0.99, and xB = 0.01. Also let the reflux ratio be r = 3.5.
- Make a yx plot for propylene and n-propane including the 45-degree line.
- Perform a graphical McCabe-Thiele analysis of this C3 splitter.
- How many total stages are there in this distillation column?
- Where is the feed stage?
- Calculate the boil-up ratio and make sure it is consistent with that shown on your McCabe-Thiele diagram.
- How much heat must be supplied to the reboiler for this separation to be performed as you have designed it?
- What is the amount of cooling that needs to be supplied to the condenser?
- What are the liquid and vapor flow rates in the rectifying and stripping sections of the distillation column?
- How many more stages would be required if we wanted product compositions of xD = 0.999 and xB = 0.001?
- What would be the corresponding change in r, s, QR, and QC?